Subarea 2: Regeneration and Homeostasis of Organs in Aging
The main goal of Subarea 2 is to identify cellular and molecular pathways used to ensure effective organ maintenance and repair, and to unravel the mechanisms of their deterioration during aging. While stem cells are important for organ homeostasis, this Subarea does not per se directly addresses stem cell aging but rather focusses on the following focus areas:
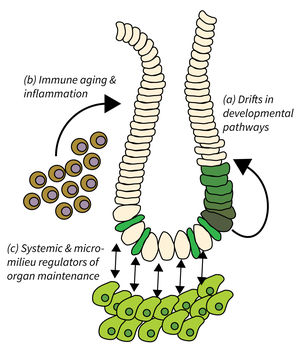
- Drifts in developmental pathways limiting organ maintenance in aging,
- Immune aging and inflammation, and
- Systemic and micro-milieu regulators of organ maintenance, regeneration, and disease development.
Research focus of Subarea 2
Organ maintenance is regulated by local and systemic factors, which are subject to aging-associated changes. Research of Subarea 2 focuses on the following research areas: a) Genetic and epigenetic modulation of developmental pathways has been shown to contribute to progressive aging and disease. It is critical to delineate mechanisms and consequences of aging-associated drifts to better understand organ maintenance during aging. b) Immunoaging and chronic inflammation elicits negative effects through reduced immune surveillance and aberrant organ repair and maintenance; all of which contributes to the evolution of organ pathologies and diseases during organismal aging. c) Furthermore, aging-associated alterations in systemic and extracellular factors derived from metabolic changes, microbiota alterations, chronic inflammation, senescent, or damaged cells might impinge on disease development and tumor initiation.
Publications
(since 2016)
2021
- Analysis of microRNA expression reveals convergent evolution of the molecular control of diapause in annual fish
Barth E, Baumgart M, Dolfi L, Cui R, Groth M, Ripa R, Savino A, R.Valenzano D, Marz M, Cellerino A
Research Square 2021, https://doi.org/10.21203/rs.3.rs - Merlin dependent intra- and intercellular signaling network alterations in NF2 disease
Carlstedt A
Dissertation 2021, Jena, Germany - Brigatinib causes tumor shrinkage in both NF2-deficient meningioma and schwannoma through inhibition of multiple tyrosine kinases but not ALK.
Chang LS, Oblinger JL, Smith AE, Ferrer M, Angus SP, Hawley E, Petrilli AM, Beauchamp RL, Riecken LB, Erdin S, Poi M, Huang J, Bessler WK, Zhang X, Guha R, Thomas C, Burns SS, Gilbert TSK, Jiang L, Li X, Lu Q, Yuan J, He Y, Dixon SAH, Masters A, Jones DR, Yates CW, Haggarty SJ, La Rosa S, Welling DB, Stemmer-Rachamimov AO, Plotkin SR, Gusella JF, Guinney J, Morrison H, Ramesh V, Fernandez-Valle C, Johnson GL, Blakeley JO, Clapp DW, Synodos for NF2 Consortium
PLoS One 2021, 16(7), e0252048 - Genome-wide functional CRISPR-Cas9 screening reveals key roles of GOLT1A and GOLT1B in the FLI-06-mediated secretion block
Cramer P
Dissertation 2021, Jena, Germany - Merlin cooperates with neurofibromin and Spred1 to suppress the Ras-Erk pathway.
Cui** Y, Ma L, Schacke S, Yin JC, Hsueh YP, Jin H, Morrison** H
Hum Mol Genet 2021, 29(23), 3793-806 ** co-corresponding authors - GMPPA defects cause a neuromuscular disorder with α-dystroglycan hyperglycosylation.
Franzka P, Henze* H, Jung* MJ, Schüler SC, Mittag S, Biskup K, Liebmann L, Kentache T, Morales J, Martínez B, Katona I, Herrmann T, Huebner AK, Hennings JC, Groth S, Gresing LJ, Horstkorte R, Marquardt T, Weis J, Kaether C, Mutchinick OM, Ori A, Huber O, Blanchard V, von Maltzahn J, Hübner CA
J Clin Invest 2021, 131(9), e139076 * equal contribution - High Yap and Mll1 promote a persistent regenerative cell state induced by Notch signaling and loss of p53.
Heuberger J, Grinat J, Kosel F, Liu L, Kunz S, Vidal RO, Keil M, Haybaeck J, Robine S, Louvard D, Regenbrecht C, Sporbert A, Sauer S, von Eyss B, Sigal M, Birchmeier W
Proc Natl Acad Sci U S A 2021, 118(22), e2019699118 - Isolation, ‘omics characterization and organotypic culture of alveolar type II pulmonary epithelial cells
Li* H, Schütte* M, Bober* M, Kroll T, Frappart L, Bou About G, Lin YC, Sorg T, Herault Y, Wierling C, Rinner O, Lange Bodo MH, Ploubidou A
bioRxiv 2021, https://doi.org/10.1101/2021.08. * equal contribution - The LIM domain protein nTRIP6 modulates the dynamics of myogenic differentiation.
Norizadeh Abbariki T, Gonda Z, Kemler D, Urbanek P, Wagner T, Litfin M, Wang ZQ, Herrlich P, Kassel O
Sci Rep 2021, 11(1), 12904 - Stimulation of Non-canonical NF-κB Through Lymphotoxin-β-Receptor Impairs Myogenic Differentiation and Regeneration of Skeletal Muscle.
Schmidt M, Weidemann A, Poser C, Bigot A, von Maltzahn J
Front Cell Dev Biol 2021, 9, 721543