CF Imaging
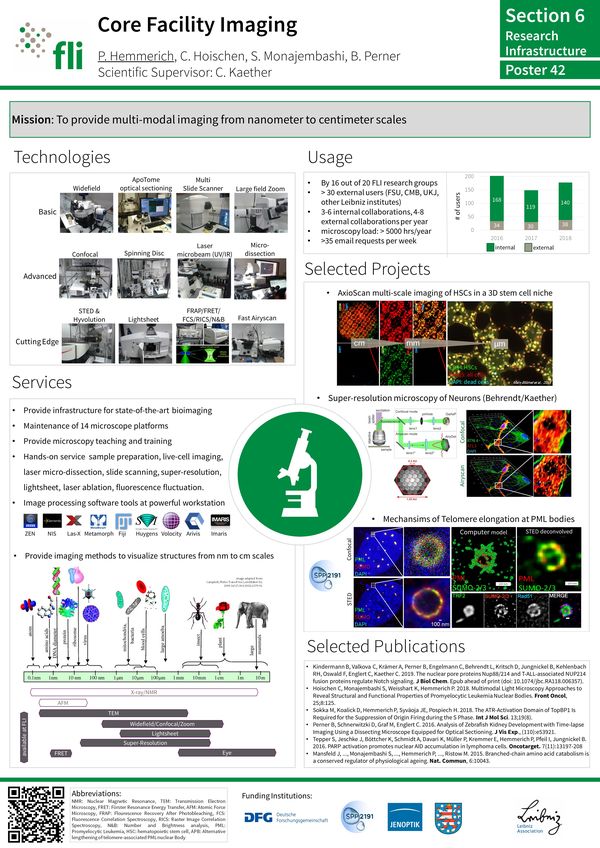
The Imaging Facility provides the infrastructure to support state-of-the-art microscopy at the FLI. Currently, the Facility operates and maintains several widefield and confocal microscopes, a Lightsheet and a STED superresolution microscope.
The facility staff provides introduction into microscopy techniques and service for FLI researchers and, upon arrangement, also for external users.
The Imaging Facility provides the infrastructure to support state-of-the-art microscopy at the FLI. Currently, the Facility operates and maintains several widefield, confocal, lightsheet (STED-) superresolution, microdissection, optical tweezer and multi-slide scanning microscopes.
The facility staff provides introduction into microscopy techniques and service for FLI researchers and, on a collaborative basis, also for external users.
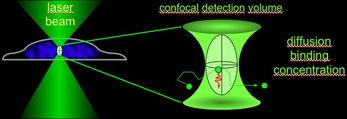
The Facility is specialized in live cell microscopy to monitor molecule and cellular dynamics in space and time. Several fluorescence fluctuation microscopy (FFM) techniques with single-molecule resolution have been established including FRAP, FCS, FCCS, FLIM, FRET, and RICS.
Equipment and techniques available:
- LSM/ConfoCor3 (FRET, FRAP, FCS, FCCS, RICS and time-lapse)
- Airyscan (multi-fluorescence, rapid live-cell imaging, superresolution)
- PALM Microdissection/Optical Tweezer
- Apotome optical sectioning (Widefield, brightfield,quasi-confocal fluorescence microscopy; colocalization analysis, 2D, 3D, distance measurements, tiling of tissue sections)
- Multi-Slide Scanners (up to 100
- Several Axiovert 200 ApoTomes; 2x inverse, 3x upright (optical sectioning with structured illumination)
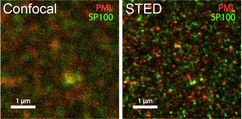
- TCS SP8 STED-3X/FALCON (Stimulated emission depletion super-resolution microscopy; FLIM)
- Lightsheet Z1 (live imaging of whole organisms in 3D over time).
- Image quantitation (Huygens, Imrais, Arivis, Metamorph)
Contact

Peter Hemmerich
CF Manager
+49 3641 65-6262
imaging@~@leibniz-fli
Christoph Kaether
Scientific Supervisor
christoph.kaether@~@leibniz-fli.de
FLI's Imaging Facility is part of the German BioImaging Network (GERBI).
- Live-cell analyses of chromatin complex assembly
- Superresolution imaging of supramolecular cellular complexes
- Live-cell imaging of intracellular vesicle transport
- Live-cell imaging of dendritic cells
- Confocal imaging of mouse tissues and 3D reconstruction
- Immunofluorescence localization of molecules in cultured cells and tissue samples
Internal users:
Download (pdf) User information on the Imaging Facility
External users:
Please inquire.
Team*
| Name | Phone | Position | |
|---|---|---|---|
| Peter Hemmerich | +49 3641 656262 | Core Facility Manager | |
| Christian Hoischen | +49 3641 656305 | Staff Scientist | |
| Birgit Perner | +49 3641 656749/6493/6409 | Staff Scientist | |
| Tobias Ulbricht | +49 3641 656260 | Research Engineer | |
| Marianne Koch | +49 3641 656168 | Technical Assistant | |
| Christoph Kaether | +49 3641 656230 | Scientific Supervisor |
* incomplete due to Data protection
For publications of individual CF members: publications search