Subarea 5: Computational and Systems Biology of Aging
Subarea 5 focuses on the development of methods to analyse and understand complex biological systems. This work includes the design of computer algorithms and biostatistical approaches as well as the development of novel Omic strategies (i.e. genomics/epigenomics, transcriptomics, proteomics, and metabolomics) to study aging and aging-related diseases. According to the FLI, due to the Subarea's expertise in computational data analysis, it is deeply interconnected with all other Subareas. The Subarea hosts two critical core facilities (Life Science Computing, Proteomics) and provides consulting services in statistics. Furthermore, it organizes courses on data analysis and statistics.
The research is defined by five focus areas:
- Mapping extrinsic and intrinsic factors influencing stem cells during aging,
- Integration of spatiotemporal proteomics and transcriptomics data,
- Comprehensive evaluation of qualitative and quantitative expression changes,
- Identification and analysis of epigenomic alterations during aging and age-related diseases, and
- Network analysis of genomic, transcriptomic and epigenomic alterations during aging.
Research focus of Subarea 5.
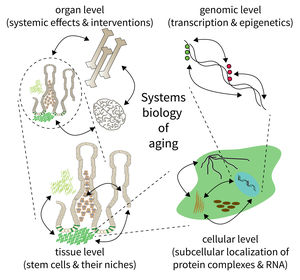
The biology of aging can be viewed as a multilayered array of networks at the level of organs, cells, molecules, and genes. The FLI wants to meet this complexity by establishing the new Subarea on “Computational and Systems Biology of Aging”. The overall goal is to interconnect research at different scales, taking place in Subareas 1-4 of the Institute’s research program. The new group on Systems Biology will integrate data from networks at multiple scales and will thus point to mechanisms and interactions that would not be seen in unilayer approaches.
Publications
(since 2016)
2021
- Tnfaip2/exoc3-driven lipid metabolism is essential for stem cell differentiation and organ homeostasis.
Deb S, Felix DA, Koch P, Deb MK, Szafranski K, Buder K, Sannai M, Groth M, Kirkpatrick J, Pietsch S, Gollowitzer A, Groß A, Riemenschneider P, Koeberle A, González-Estévez** C, Rudolph** KL
EMBO Rep 2021, 22(1), e49328 ** co-corresponding authors - Abundance and size of hyaluronan in naked mole-rat tissues and plasma.
Del Marmol D, Holtze S, Kichler N, Sahm A, Bihin B, Bourguignon V, Dogné S, Szafranski K, Hildebrandt TB, Flamion B
Sci Rep 2021, 11(1), 7951 - Mapping sites of carboxymethyllysine modification on proteins reveals its consequences for proteostasis and cell proliferation
Di Sanzo S
Dissertation 2021, Jena, Germany - Mapping protein carboxymethylation sites provides insights into their role in proteostasis and cell proliferation.
Di Sanzo* S, Spengler* K, Leheis A, Kirkpatrick JM, Rändler TL, Baldensperger T, Dau T, Henning C, Parca L, Marx C, Wang ZQ, Glomb MA, Ori** A, Heller** R
Nat Commun 2021, 12(1), 6743 * equal contribution, ** co-senior authors - Mice Are Not Humans: The Case of p53.
Fischer M
Trends Cancer 2021, 7(1), 12-4 - GMPPA defects cause a neuromuscular disorder with α-dystroglycan hyperglycosylation.
Franzka P, Henze* H, Jung* MJ, Schüler SC, Mittag S, Biskup K, Liebmann L, Kentache T, Morales J, Martínez B, Katona I, Herrmann T, Huebner AK, Hennings JC, Groth S, Gresing LJ, Horstkorte R, Marquardt T, Weis J, Kaether C, Mutchinick OM, Ori A, Huber O, Blanchard V, von Maltzahn J, Hübner CA
J Clin Invest 2021, 131(9), e139076 * equal contribution - Altered Glycosylation in the Aging Heart.
Franzka P, Krüger L, Schurig MK, Olecka M, Hoffmann S, Blanchard V, Hübner CA
Front Mol Biosci 2021, 8, 673044 - The Hematopoietic Bone Marrow Niche Ecosystem.
Fröbel J, Landspersky T, Percin G, Schreck C, Rahmig S, Ori A, Nowak D, Essers M, Waskow C, Oostendorp RAJ
Front Cell Dev Biol 2021, 9, 705410 - Identifying Cell-Type-Specific Metabolic Signatures Using Transcriptome and Proteome Analyses.
Gebert N, Rahman S, Lewis CA, Ori** A, Cheng** CW
Curr Protoc 2021, 1(9), e245 ** co-corresponding authors - Alternative Animal Models of Aging Research
Holtze S, Gorshkova E, Braude S, Cellerino A, Dammann P, Hildebrandt TB, Hoeflich A, Hoffmann S, Koch P, Terzibasi Tozzini E, Skulachev M, Skulachev VP, Sahm A
Front Mol Biosci 2021, 8, doi: 10.3389/fmolb.2021.660959.