Assets of the last resort: When Stem Cells are Aging
Many cells in the human body are able to divide themselves, thus helping tissues and organs to regenerate, e.g. if wounds are healing. But this regenerative capability is limited.
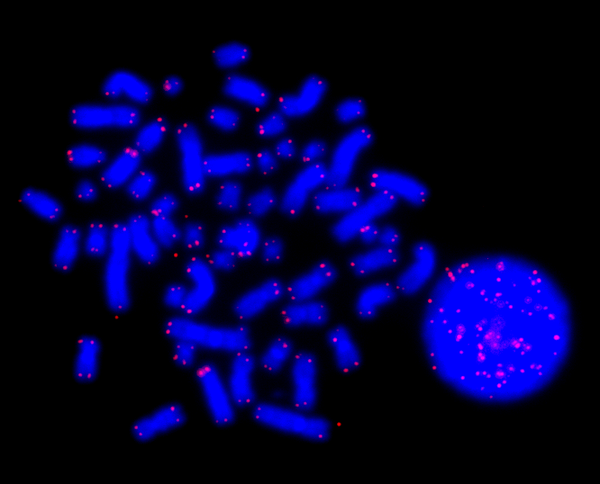
One reason for the reduced regeneration during aging is that our stem cells, so-to-speak the mother cells of all types of cells, age as well. To defend our DNA from damage due to cell division, it has specialized protective caps, so-called telomeres that shorten with every cell division. Enzyme Telomerase is in charge of renewing these protective caps – but only for certain cell types. For all others, genetic information is lost or damaged when after a certain number of cell divisions the telomeres are gone, leading to cell death (and, thus, to organ dysfunctions) or to the proliferation of “wrong” cells, which in consequence may lead to cancer.
Many stem cells try to avoid these damages by “sleeping”. They stay in the so-called state of “quiescence” and are only activated in case of severe tissue injury when a strong regeneration is needed. After they have finished their work, dividing into several sub-types of cells needed to repair and maintain a tissue, they rest and are inactive. Therefore, their telomeres are protected from shortening.
With aging, this falling-back into dormancy period is disturbed, leading to a permanent activation of stem cells and thus to the depletion of our genetic reserves. In the end, also without their protective telomeres, cell division is disturbed, altered cells can proliferate, or the stem cells can die.









